A device called the Total Artificial Heart helps some of the sickest heart-failure patients regain function — outside of the hospital — while awaiting a transplant.
7:00 AM
Author |

It sounds like a byproduct of science fiction: a synthetic, battery-powered device designed to replace the work of the body's most crucial organ.
MORE FROM MICHIGAN: Sign up for our weekly newsletter
But the equipment, known as the Total Artificial Heart, offers a lifeline for some of the most severe heart-failure cases.
"That's what the evolution of technology can do," says Jonathan Haft, M.D., a cardiac surgeon at the University of Michigan Health System. "You're actually removing the heart and replacing it."
Haft pointed to his patient Stan Larkin, pictured above, a 25-year-old with a rare form of cardiomyopathy who lived for 555 days — outside of the hospital — using a Total Artificial Heart before receiving a heart transplant at UMHS in May. "He's absolutely thriving now."
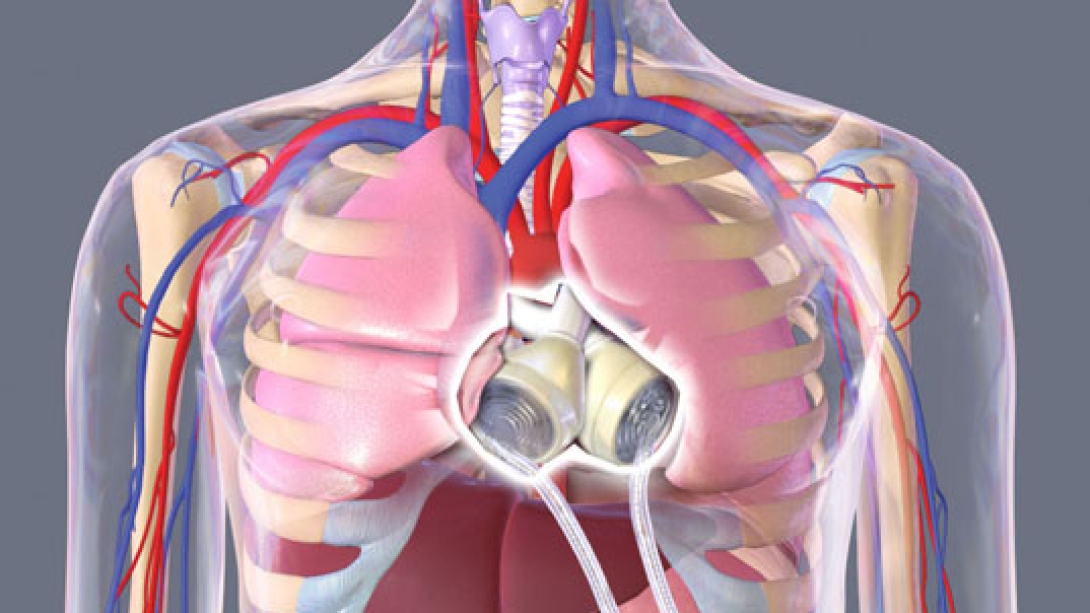
The internal mechanics, which replace both ventricles and four heart valves, are regulated by two tubes that connect to a portable, 13.5-pound external air pump called the Freedom driver. (Until the Freedom driver's approval by the Food and Drug Administration two years ago, most recipients had to rely on a hulking, 418-pound driver the size of a washing machine, which kept them hospitalized full time.)
Launched in 1969 and evolving since the 1980s, the devices aren't for everyone.
Only the left ventricle is affected in about 85 to 90 percent of patients with advanced end-stage heart failure — which qualifies them for the smaller and less-invasive pump known as a left ventricular assist device (LVAD).
For those facing biventricular failure, however, a Total Artificial Heart is "the only real option for them," Haft says.
Compared with the LVAD, implementation of the Total Artificial Heart is far less common.
Between one and seven such surgeries for the latter occur at UMHS each year, says Haft, who explained the particulars in his own words.
7 things to know about Total Artificial Hearts
They require surgery: The operation is not too dissimilar to a heart transplant; somewhere in the neighborhood of four to five hours. It can be complicated or challenged by prior operations. If someone has been in the hospital for a long period of time, the recovery could take months.
They are large: It's noticeably bigger than a normal human heart. Patients have to undergo a CT scan to determine if there's enough room. This particular device has a certain size requirement in the chest cavity. For normal-sized people with chronic heart failure, usually it fits.
SEE ALSO: A Congenital Condition that Lead to 'Surprise' Heart Surgery
They allow mobility: The Freedom driver has a battery, so patients can be relatively untethered for a period of about two to three hours. Patients can use backup batteries if they have to leave the house for longer amounts of time. Of course, they can plug into an outlet whenever needed.
They can be unwieldy: The external controller is large and heavy. It's a burden for people to have to carry all the time. It's very, very noisy. Another limitation is the two large hoses that exit the body can be cumbersome and potentially painful.
They carry risks: The No. 1 challenge is infection … (via) a piercing of the skin (for the hoses) where germs can get in. With a huge amount of interface between blood and foreign surface, there are hazards of clots forming on the inside of the body. And if a patient is taking blood thinners, they can develop bleeding complications.
They are temporary: There are challenges with long-term support, so it's intended for transplant-eligible patients or those we intend to provide a transplant once they become eligible. There are no specific time limits for the device, and the durability and risk of complications over time remain unknown.
They are evolving: The 70cc pump (the volume of blood that fills the device) has size limitations. You can't implant it into somebody who is very small or in people with normal-sized hearts. An investigational 50cc pump currently in clinical trials could increase eligibility for small people and for women.

Explore a variety of healthcare news & stories by visiting the Health Lab home page for more articles.

Department of Communication at Michigan Medicine
Want top health & research news weekly? Sign up for Health Lab’s newsletters today!





