Gifts in memory of Chad Carr, grandson of former Michigan football coach Lloyd Carr, will help Michigan scientists to fight the deadly DIPG tumor Chad battled.
8:00 AM
Author |

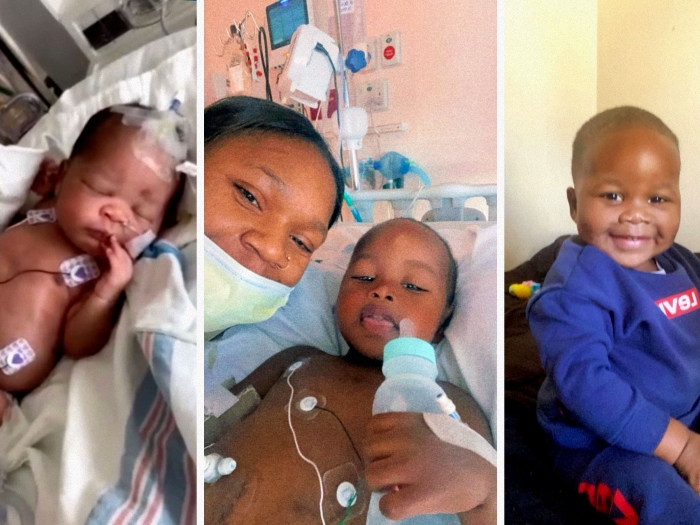
As Chad Carr fought brain cancer in the hospital wing named for his grandfather at C.S. Mott Children's Hospital, he united and galvanized the Michigan community.
Chad, the grandson of former Michigan football coach Lloyd Carr, died at age 5 in 2015, 15 months after his diagnosis, but not before his story captured national attention for brain cancer research — and spawned the #ChadTough movement.
SEE ALSO: 'Superhero' Brothers Bonded by Rare XMEN Disease
Today, his memory lives on at Mott through research funded by a stream of donations to the ChadTough Fund at the University of Michigan and a recent research grant in Chad's name from The V Foundation for Cancer Research.
To help drive new treatments for the cancer that took Chad's life, Mott scientists will use the funds to study how the highly aggressive and inoperable DIPG brain tumor develops.
Brain tumors are the leading cause of death from childhood cancer, and the most devastating type is DIPG (diffuse intrinsic pontine glioma), which stems from the region of the brain that controls breathing and heart rate.
More than 90 percent of children diagnosed with DIPG die within 18 months.
"Outcomes for DIPG are dismal and unacceptable. We have made great strides in treating and curing many childhood cancers, but research over the last 25 years has failed to significantly improve the prognosis for children with this brain cancer," says Valerie Opipari, M.D., a pediatric oncologist and chair of the Department of Pediatrics and Communicable Diseases at C.S. Mott Children's Hospital.
"There is a desperate need to develop entirely new treatment approaches to fight this heartbreaking disease. Chad's brave fight has inspired our community to rally together and be a part of finding the cure."
There is no other place where we would rather invest our time and efforts in finding a cure for DIPG and keeping Chad's legacy alive than Mott Children's Hospital.Tammi Carr
About the research
Chad's story prompted hundreds of donations from throughout the country to U-M's ChadTough Fund, established in 2014.
SEE ALSO: Doctors Unleash New Weapon to Fight Pediatric Neuroblastoma
The fund is already contributing to several important initiatives at Michigan, including research involving Chad's own DIPG tumor tissue.
This study, led by Mott pediatric oncologist Rajen Mody, M.D., includes extensive genetic analysis of Chad's brain tissue to explore the biology of DIPG and better understand genetic changes within different regions of the tumor and how it compares with normal tissue. Through this work, researchers can potentially better understand genetic changes driving tumor growth, which may aid in designing future therapies.
"We are a Michigan family through and through," Chad's mother, Tammi Carr, says. "There is no other place where we would rather invest our time and efforts in finding a cure for DIPG and keeping Chad's legacy alive than Mott Children's Hospital."
The V Foundation grant in Chad's memory will also allow Mott to devote new resources to advanced immunotherapy research. Immunotherapy, which stimulates the immune system to work harder or smarter to attack tumors, has helped some patients with resistant and difficult-to-treat cancers, including lymphoma and neuroblastoma.
The major challenge to using immunotherapy in DIPG and other cancers, however, is that the disease creates an environment that hides the cancer cells from the immune system, preventing it from recognizing and fighting the cancer. Scientists hypothesize that specific mutations in DIPG tumor cells are to blame.
A team led by Luigi Franchi, M.D., Ph.D., an assistant professor of pediatrics in hematology and oncology at Mott, will study cell lines from DIPG tumors to test which ones may be responsible for blocking immune signaling in order to identify better drug targets to treat DIPG.
Over the next five years, the U-M teams will generate and study subjects with DIPG tumors, collect data and perform drug screening and testing of the newest, most promising therapies available.
Related work at U-M
Other DIPG-related research projects underway at Michigan that pediatric brain cancer research donations support include:
Development of the first genetically engineered model of DIPG. One of the reasons scientists haven't made significant advances in treatment is that there haven't been any good models of the disease to study.
But now, a team of U-M specialists led by Maria G. Castro, Ph.D., has developed the first animal model of pediatric high-grade gliomas, or malignant brain tumors. The mice carry the genetic mutations found in human tumors, and develop tumors that closely resemble those in children and adolescents — allowing researchers to study what drives DIPG and develop and test potential new drug treatments.
Next-generation genomic sequencing. Researchers are combing through patients' entire genome, and to personalize treatment, zeroing in on genetic mutations that make a child's cancer unique and cause their tumor to grow.
This work at Michigan, led by genomics expert Arul Chinnaiyan, M.D., Ph.D., of the U-M Comprehensive Cancer Center, and Mody, will expand the comprehensive genomic analysis of pediatric tumors to find new treatable targets and precision medicine treatments for resistant cancers such as DIPG. This includes analyzing Mott patients and providing screening for other regional children's hospitals as well.

Explore a variety of healthcare news & stories by visiting the Health Lab home page for more articles.

Department of Communication at Michigan Medicine
Want top health & research news weekly? Sign up for Health Lab’s newsletters today!