A rare case of right-sided congenital diaphragmatic hernia encouraged a Michigan Medicine team to expand access to an experimental, life-saving procedure.
5:00 AM
Author |
Erin Perrone, M.D., will never forget her first meeting with Kirsten and Mike VanDerMeulen. And now, three years later, that meeting may help change the lives of many families in the Midwest to come.
The Hudsonville, Mich., couple visited the Fetal Diagnosis and Treatment Center at Michigan Medicine when Kirsten was 28 weeks pregnant for a consultation on a congenital diaphragmatic hernia (CDH). A CDH is a hole in the diaphragm of unborn babies that allows abdominal organs to move into the chest and restrict lung development. It's a rare, complex abnormality in fetal development that can cause lifelong complications or, in more severe cases, death.
The VanDerMeulen's case was particularly rare. The unborn baby — now Jordan, who turns three in January — had right-sided CDH, which is present in only about 15% of cases. Jordan's low lung volume was concerning to Perrone, a pediatric surgeon who specializes in pediatric and fetal surgery.
LISTEN UP: Add the Michigan Medicine News Break to your Alexa-enabled device, or subscribe to our daily updates on iTunes, Google Play and Stitcher.
"Dr. Perrone told us that [the hernia] was more severe than originally diagnosed and … that there was very little left lung tissue and very little, if any, on the right lung," Kirsten says. "She said it's not hopeless, but it is very, very concerning. We talked about comfort care and different options for Jordan. That was the first time we felt our hope for Jordan's survival deflate."
Finding FETO
That night, Perrone couldn't put the heartbreaking case out of her mind. She started making calls, finding colleagues at the Baylor College of Medicine and Texas Children's Hospital in Houston who were treating severe CDH cases with an experimental in utero surgery called fetal endoscopic tracheal occlusion, or FETO for short.
During the surgery, a balloon is inserted into the fetus' trachea to help expand the small lungs and help them to grow. Mother and baby are carefully monitored, typically for a month or more, at which point the balloon is removed. It's a newer procedure, only offered at about 10 institutions in the country. At the time, Houston was the only center offering FETO for right-sided CDH. It doesn't always work, and evidence is limited for babies with right sided CDH because it's so rare.
But Perrone thought the VanDerMeulens had a shot.
Connecting the dots
Perrone called the family the day after her initial consult and asked permission to send their information to Texas. She said they had to act fast: the FETO balloon must be inserted by 29 weeks and 6 days of pregnancy, and Kirsten was entering her 29th week. It's still an experimental procedure with strict requirements, so a thorough evaluation was needed to determine if Kirsten was even a candidate.
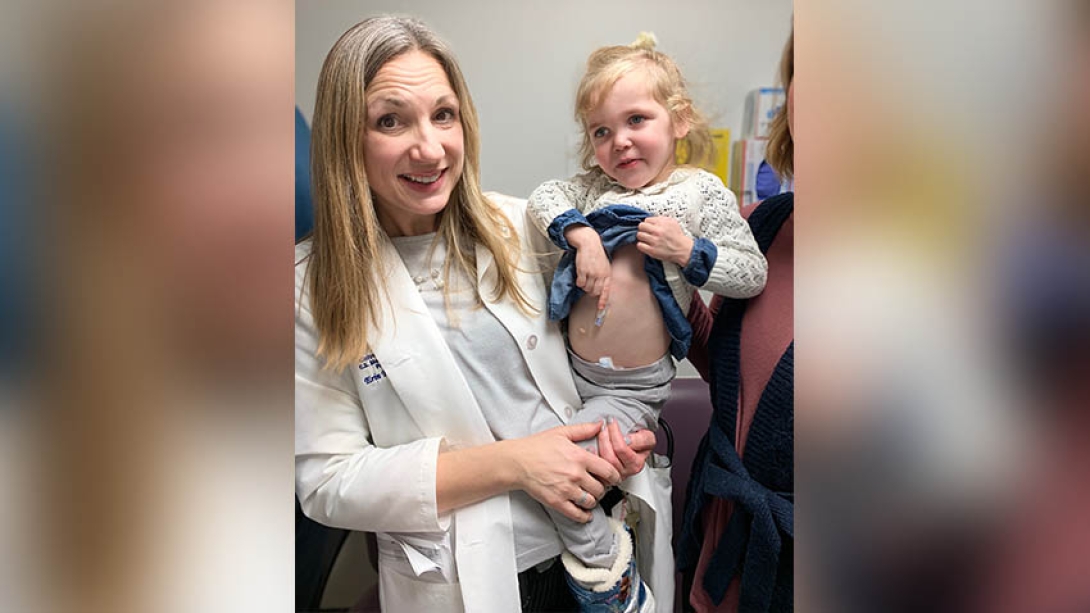
From there, it was a whirlwind. The VanDerMeulens traveled to Houston and had the procedure within the week. Perrone and her Michigan Medicine colleague, Marcus Jarboe, M.D., flew to Texas to observe the surgery and support the family.
"The balloon was in just shy of four weeks. That was the goal," Kirsten says. "And then the goal was to stay pregnant as long as possible."
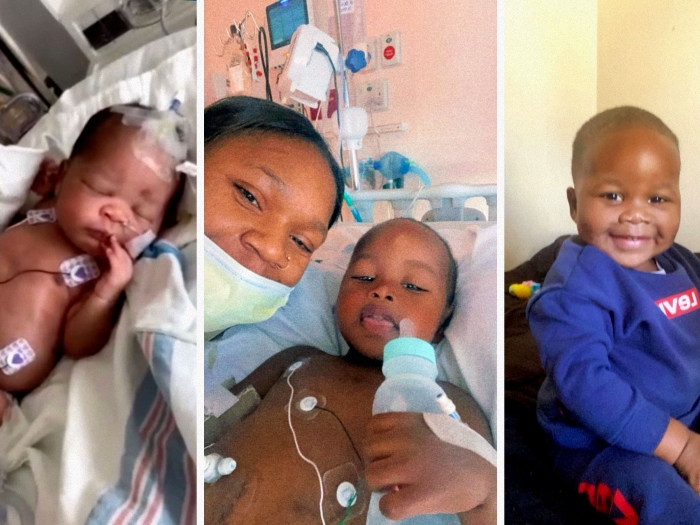
FETO helped improve Jordan's lung development, and she was born at 36 weeks and 3 days. Jordan was rushed to the NICU at Texas Children's, where she would stay for 64 days, taking liquid nutrition and receiving breathing support. At just three days old, she had her hernia repaired.
On the mend
Back in Michigan, the VanDerMeulens resumed care at the C.S. Mott Children's Hospital. Jordan continued to use oxygen and Dr. Perrone placed a gastrostomy tube for feeding. Because of her low lung strength and compromised immune system, the family was almost entirely home-bound during Jordan's first year of life.
Today, Jordan continues to be followed closely by a long-term, multidisciplinary CDH clinic team at U-M to ensure proper growth and development. She was recently able to have her gastrostomy removed since her feedings improved orally over the past year. And for the most part, she's a healthy toddler and big sister to her brother Ryan, 1.
MORE FROM MICHIGAN: Sign up for our weekly newsletter
"She loves to run and play, and she talks — she speaks very well," Kirsten says. "We thought developmental delays would be part of her life, but she's done so, so well. She's impressed her medical teams and really surpassed what a lot of people expected for her. To see where she started and where she is now, it's astounding."
Spreading hope through a new clinical trial
As the birthplace of extracorporeal membrane oxygenation (ECMO) and other pioneering treatments for CDH, Michigan Medicine has a long history of innovation for CDH patients; now they are poised to offer fetal treatment. While treating Jordan, Perrone and others started working to bring FETO to Michigan. She had other patients who could've qualified for the procedure, but were unable to travel due to a variety of reasons. Since she didn't want to see any other eligible families struggle to get the care they desired, Perrone and her colleagues decided to launch an FDA approved clinical trial for FETO at Michigan Medicine (HUM00159792). The trial's currently enrolling pregnant women carrying babies with severe CDH, including those with right sided CDH, like Jordan.
"To see the FETO trial come to Mott and see them accept right-sided cases like Jordan's is such an honor," says Kirsten. "We will raise her telling her that you can spread hope to other people."
Says Mike, "I believe we would not be in the position we are in today, or that Jordan would even be here, if Dr. Perrone hadn't cared so much about Jordan and her situation and Kirsten's pregnancy. That's meant more than we could even begin to summarize. … It really makes us feel good knowing that Jordan's story can have an impact on other families."
Adds Perrone, "For a lot of families with this diagnosis prenatally, it comes as a shock and is very difficult. The one thing that this trial does is offer hope for families that didn't have it beforehand. Our team can help offer hope and options."
In addition to Perrone, the clinical trial team includes co-principal investigator Marjorie C. Treadwell, M.D. Other co-investigators include: Deborah Berman, M.D.; Alissa Carver, M.D.; Marcus Jarboe, M.D.; and George Mychaliska, M.D.

Explore a variety of healthcare news & stories by visiting the Health Lab home page for more articles.

Department of Communication at Michigan Medicine
Want top health & research news weekly? Sign up for Health Lab’s newsletters today!