A new, minimally invasive option is now available for patients with severe aortic stenosis who are at intermediate risk for open-heart surgery.
7:00 AM
Author |
In the past, severe aortic stenosis patients with moderate risk had one option: open-heart surgery.
MORE FROM MICHIGAN: Sign up for our weekly newsletter
A minimally invasive procedure known as transcatheter aortic valve replacement (TAVR) was developed six years ago. But it was an option only for high-risk patients who were not candidates for surgical aortic valve replacement (SAVR) to treat their condition.
Until now: The U.S. Food and Drug Administration has approved the use of two TAVR systems, Edwards Sapien and Medtronic CoreValve, for symptomatic severe aortic stenosis patients at moderate risk for surgery.
"Moderate-risk patients now have a choice," says P. Michael Grossman, M.D., an interventional cardiologist at the University of Michigan Frankel Cardiovascular Center.
The FDA decision is based in part on findings from the SURTAVI (Surgical Replacement and Transcatheter Aortic Valve Implantation) trial, which evaluated the safety and efficacy of TAVR in severe aortic stenosis patients at intermediate risk for surgery.
Michigan Medicine was a leading center in the trial.
Opening new doors
Grossman thinks this development will significantly impact the future of TAVR — namely, the ability to offer the treatment to a wider population.
Unlike a SAVR procedure, which involves opening the chest to replace a patient's aortic valve with a mechanical or tissue valve, the minimally invasive TAVR approach delivers a fully collapsible replacement valve via catheter through a large artery in the groin or chest.
SEE ALSO: Staying Alive with the Help of TAVR and Sisterly Love
"Patients who were too ill for open-heart surgery several years ago are today routinely treated with TAVR," Grossman says.
The procedure requires two small incisions. Once the new valve is expanded, it pushes the old valve leaflets out of the way and the tissue in the replacement valve takes over the job of regulating blood flow.
TAVR patients often recover much quicker than SAVR patients, Grossman notes, which might allow them to leave the hospital and start cardiac rehabilitation sooner — timing that can help improve quality of life.
Full evaluation is critical
Although many patients might prefer a less invasive approach to treating aortic stenosis, a detailed review must come first.
"All patients with severe aortic stenosis symptoms should be fully evaluated at a center of excellence for treatment options, which may include TAVR," Grossman says. "A careful evaluation needs to be done to make the best choice possible for each patient."
Still, he adds, SAVR remains the preferred method for many patients: "It has saved and prolonged tens of thousands of lives over the span of many decades. Surgical techniques have evolved, and the valve technology is excellent with proven durability."
Moderate-risk patients, many of whom have no other medical issues, will nonetheless need to work with their health care team in developing a plan that takes a recipient's lifespan into consideration.
The reason: "We have limited data on long-term outcomes for these patients," Grossman says. "However, the data for high-risk patients undergoing TAVR shows the procedure to be durable for several years."
Expanding, testing TAVR
The TAVR team at Michigan Medicine has performed more than 900 TAVR procedures to date — the most of any health system in the state, making it one of the top programs in the country in volume.
And with the recent opening of the Frankel Cardiovascular Center Robert and Ann Aikens Hybrid Suite, TAVR procedures are being done in greater numbers than ever — a volume that is poised to increase with the recent FDA recommendations.
The Frankel Cardiovascular Center also is one of only two centers in Michigan invited to participate in a clinical trial offering TAVR for low-risk patients, the nation's first study of a TAVR device in this population. These patients, whose aortic stenosis would typically receive an open-heart SAVR, can now be considered a candidate for a TAVR procedure.
Find out more about Cardiovascular Research Studies at the Frankel Cardiovascular Center.
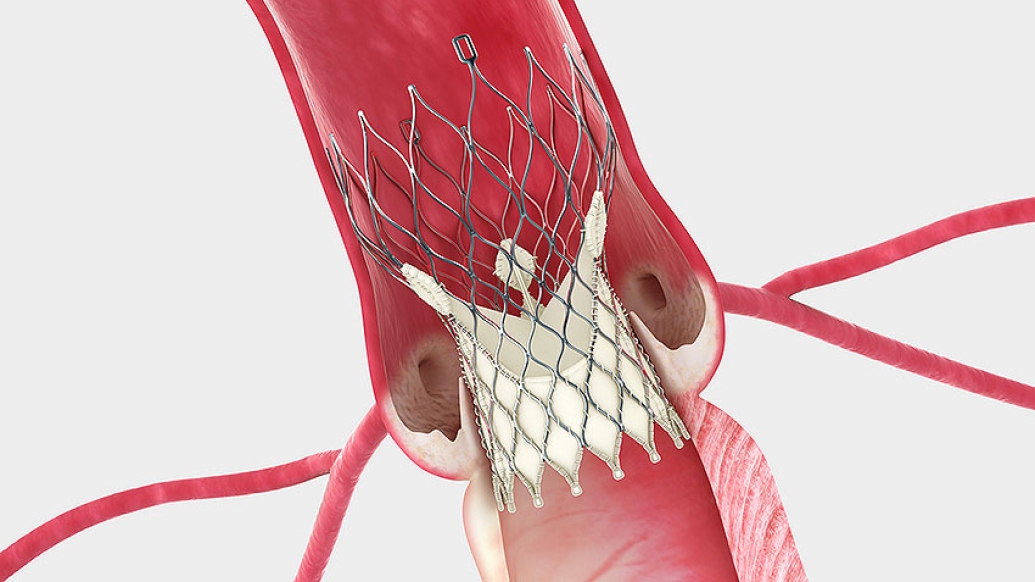
Image: The Medtronic CoreValve® System sits within the same area as the native valve. (Courtesy Medtronic)

Explore a variety of healthcare news & stories by visiting the Health Lab home page for more articles.

Department of Communication at Michigan Medicine
Want top health & research news weekly? Sign up for Health Lab’s newsletters today!